- Announced plans to advance FT-104, its novel psychedelic compound, to clinic for Treatment-Resistant Depression and Postpartum Depression as the lead indications.
- Initiated a new pipeline research program focused on discovering novel psychedelics with a reduced cardiovascular risk profile compared to classic psychedelics (the “FT-200 Group”) and filed a provisional patent in connection to the composition of matter for the first molecule identified in the FT-200 Group.
- Continued to invest in best-in-class clinical infrastructure, with nine clinics currently in operation and nine locations under construction or about to commence construction.
- Commenced trading on the NASDAQ Global Select Market (“NASDAQ”) under the ticker symbol “FTRP”.
- As at September 30, 2021, Field Trip had approximately $88 million in unrestricted cash and cash equivalents and short-term investments.
- Commenced a strategic review of the current corporate structure to assess options to maximize the value of the drug development and therapy delivery business units.
TORONTO, Nov. 15, 2021 (GLOBE NEWSWIRE) — Field Trip Health Ltd. (TSX: FTRP; FTRP.WT; NASDAQ: FTRP) (“Field Trip“), a leader in the development and delivery of psychedelic therapies, reported its second fiscal quarter 2022 results for the three months ended September 30, 2021 and provided a business update. All results are reported under International Financial Reporting Standards (“IFRS“) and in Canadian dollars, unless otherwise specified.
Key Highlights and Recent Developments
During the quarter, Field Trip progressed its strategy of building a leading psychedelic therapy company (Field Trip Health) and continued to invest in its drug development pipeline and program expansion (Field Trip Discovery).
Field Trip Discovery
FT-104
During the quarter, it was announced that the lead indications for FT-104, Field Trip’s lead novel psychedelic compound in development (patent pending), will be Treatment Resistant Depression and Postpartum Depression. FT-104 is a novel, synthetic 5HT2A receptor agonist compound. It is currently advancing through preclinical studies with the in vivo portion completed, and final results from safety pharmacology and GLP toxicology are expected in calendar Q4 2021. To date, GLP toxicology, cardiovascular, pulmonary, and neurological safety pharmacology studies, as well as genotoxicity potential, all continue to be encouraging. In addition, final manufacturing of clinical trial material is scheduled for production to enable FT-104 to move into Phase 1 clinical trials in the first half of calendar 2022. The Company has experienced, and may continue to experience, delays in initiating Phase 1 clinical trials due to the ongoing COVID pandemic and delays at its contract manufacturing organization.
FT-200 Group: Introducing Field Trip’s Second Novel Psychedelic Program
The Company also announced the discovery of a novel molecule that, based on in vivo assay details, has the structure of classical psychedelics and has demonstrated improved selectivity for the target serotonin 2A receptor (5HT2A) relative to FT-104 and psilocybin versus off target serotonin, 5HT1A, 5HT2B and 5HT2C receptors. This is meaningful because off-target 5HT2B activity has been associated with increased risk of cardiovascular toxicity. Based on this discovery, the Company is expanding the scope of its development pipeline to focus on a new group of molecules termed the FT-200 Group, (which includes the molecule mentioned above), that have the structure of classical psychedelics, with similar potency at the 5HT2A receptor as FT-104 and psilocybin, but with reduced or the absence of activity at the off-target 5HT2B receptor. The aim of the work is that by reducing or eliminating 5HT2B activity it may allow molecules like those in the FT-200 Group to be administered more frequently, such as more chronic or chronic intermittent administration or ‘microdosing’ strategies.
“The first molecule identified in the FT-200 Group demonstrates significant promise to maintain 5HT2A activity while reducing off-target serotonin receptor activity. We are continuing to conduct preclinical work on this molecule and will explore structural analogs within the FT-200 Group to better refine and optimize this new family of substances, understand their properties better and work towards identifying a lead candidate”, said Joseph del Moral, Field Trip’s Co-founder and CEO.
On October 29, 2021 the Company filed a provisional patent application in the United States to protect the composition, as well as potential formulations and uses of the first molecule in the FT-200 group.
Field Trip Health Centres
Leveraging the growing awareness of Field Trip’s psychedelic-assisted therapy clinics, Field Trip announced on August 31, 2021 the launch of the KAP Co-operative Program (“KAP Co-op”), a program that enables eligible independent psychedelic therapists to provide ketamine-assisted psychotherapy (and, in the future, other legal, psychedelic-assisted therapies) at its Field Trip Health Centres. Field Trip also announced the launch of training programs designed to provide interested psychotherapists or other qualified mental health professionals and clinicians with access to best-in-class training on KAP.
The Company continues to invest in its clinics and during the quarter, entered into lease agreements for locations in Dallas, TX, Miami, FL and Scottsdale, AZ. Subsequent to the quarter end, the Company announced the opening of the Seattle, WA clinic, its sixth in the United States and ninth overall, along with the recent opening of a location in Fredericton, NB, and the imminent opening of a location in Vancouver, BC.
Hannan Fleiman, Field Trip’s Co-Founder and President, said, “We continue to see strong growth in our clinic business and our team is proud of the outcomes we are helping our patients achieve. We have continued to make significant investments in building out the leading platform for the delivery of psychedelic therapies and to position our clinics to be at the absolute forefront of our industry in establishing the critical infrastructure for the current and emerging psychedelic therapies with 9 clinics now in operation and 9 locations under construction or about to commence construction. Importantly, the clinics enable Field Trip to capture significant amounts of patient data on clinical outcomes which we expect to help inform future clinical development and treatment strategies.”
From a brand perspective, Field Trip continues to be a globally recognized company in the industry. During the quarter, the Company was featured in many top-tier print and broadcast media outlets, including Forbes, Vox Media, Insider, People and others, generating nearly 2 billion total potential media impressions. Field Trip’s brand presence and reach continues to generate strong website traffic and patient interest in the KAP treatments offered by the Company.
NASDAQ Listing
On July 29, 2021, Field Trip’s Common Shares commenced trading on the NASDAQ Global Select Market (“NASDAQ”) under the ticker symbol “FTRP”, providing the Company with greater access to capital. The Common Shares continue to trade in Canada on the TSX under its current symbol FTRP. Concurrent with the NASDAQ listing, Field Trip’s Common Shares ceased to be quoted on the OTCQX. The Company previously completed the process to ensure its shares are eligible for electronic clearing and settlement through the Depository Trust Company (DTC). In addition, Ronan Levy and Ellen Lubman joined the Compensation Committee and Mujeeb Jafferi and Dr. Ryan Yermus resigned as directors of the Company. Mr. Jafferi and Dr. Yermus continue to serve as Field Trip’s Chief Operating Officer and Chief Clinical Officer, respectively.
Mr. del Moral, continued, “The milestone of listing on NASDAQ during the quarter was testament to the rapid progress we have made and increased our visibility in the marketplace, improving trading liquidity and ultimately enhancing long term shareholder value as we further strengthen our leadership position in the psychedelic medicine industry.”
Strategic Review of Corporate Structure
The Company has commenced a strategic review designed to ensure that each operating unit is best positioned, optimally resourced, and focused to provide maximum long-term value to all stakeholders.
Mr. Joseph del Moral, said, “With the FT-104 nearing the clinic, the expansion of discovery efforts around our FT-200 Group, and the growing number of opportunities for Field Trip Health Centers we believe it is the correct time to review all strategic options to ensure we continue to maximize the growth potential and value of each business unit.”
The Company has engaged Bloom Burton Securities Inc. as its financial advisor in connection with the strategic review.
Financial Highlights
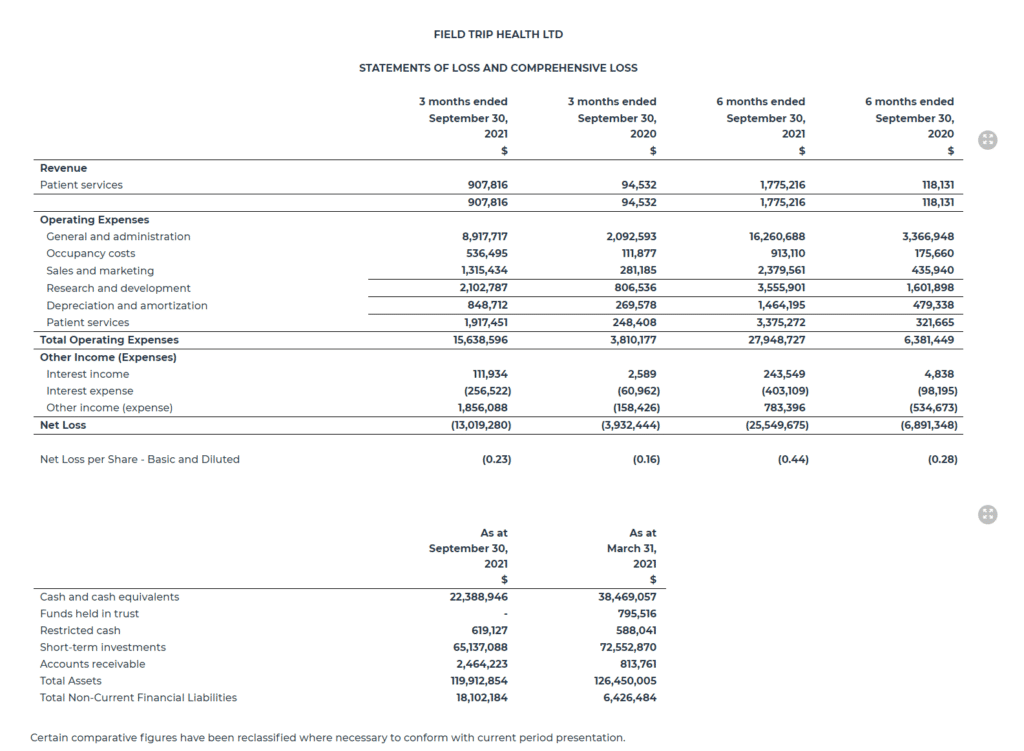
For the second fiscal quarter ended September 30, 2021, the Company earned patient services revenues of $907,816 from its Toronto, New York, Santa Monica, Chicago, Atlanta, Houston and Amsterdam clinics, an increase of $813,284 or 860% over the comparative quarter ended September 30, 2020 of $94,532 and an increase of $40,416 or 5% over the prior fiscal first quarter. The Amsterdam clinic began generating revenues in September 2021. Second fiscal quarter 2021 patient services revenues were generated from only two clinics, Toronto and New York. The modest quarter over quarter revenue increase was in part due to the COVID-19 Delta variant and seasonality associated with the slower summer months. Revenues in the first part of the third quarter indicate a clear upward trend as a result of recent process optimizations to accelerate patient on-boarding and increase clinic capacity.
Net loss for the second fiscal quarter of $13,019,280 was primarily due to total operating costs of $15,638,596, of which $2,055,890 was related to non-cash share-based compensation and $848,712 was related to non-cash depreciation and amortization. This was partially offset by a foreign exchange gain of $1,856,088. This compares with a net loss of $3,932,444 in the second fiscal quarter of 2021. The increase from the prior year primarily reflects the Company’s focus on growing the business and continued investment in its drug development pipeline and best-in-class clinic infrastructure. As the Company continues to scale, it is optimizing and streamlining the development of its psychedelic-assisted therapies.
Total operating costs in the second fiscal quarter were $15,638,596 and were comprised of the following: general and administration expenses of $8,917,717, research and development expenses of $2,102,787, patient services expenses of $1,917,451, sales and marketing expenses of $1,315,434, depreciation and amortization of $848,712 and occupancy costs of $536,495. This compares with total operating costs of $3,810,177 in the second fiscal quarter of 2021.
The difference in general and administrative expenses in the second fiscal quarter also included $596,055 in non-recurring expenses primarily related to the NASDAQ uplisting, $1,330,847 in recurring public company costs as well as non-cash share-based payments of $1,380,398.
Balance Sheet
As of September 30, 2021 Field Trip had unrestricted cash and cash equivalents, funds held in trust and short-term investments of $87,526,034.
Selected Consolidated Financial Information
The following table sets forth selected financial information derived from the Company’s unaudited condensed interim financial statements for the three months and six months ended September 30, 2021 prepared in accordance with IAS 34 in a manner consistent with the Company’s annual audited financial statements. The following information should be read in conjunction with the financial statements and management’s discussion and analysis, which are available on the Company’s website at www.fieldtriphealth.com and under the Company’s SEDAR profile at www.sedar.com.
Conference Call
The Company will conduct a conference call and webcast to discuss its results the following morning, Tuesday, November 16 at 8:30 am ET. To access the call, please dial 1-877-407-9716 (within the U.S.) or 1-201-493-6779 (outside the U.S.) and provide conference ID 13724604. A live webcast of the conference call can be accessed via the Events and Presentations section of the Field Trip Health Investor Relations website here.
For those unable to attend the live call, a telephonic replay will be available until 11:59 pm ET on Tuesday, November 30, 2021. To access the replay of the call dial 1-844-512-2921 (within the U.S.) or 1-412-317-6671 (outside the U.S.) and provide conference ID 13724604. An archived copy of the webcast will be available on the Events and Presentations section of the Field Trip Health Investor Relations website after the conclusion of the call.
About Field Trip Health Ltd.
Field Trip is a global leader in the development and delivery of psychedelic therapies. With our Field Trip Discovery division leading the development of the next generation of psychedelic molecules and conducting advanced research on plant-based psychedelics and our Field Trip Health division building centres for psychedelic therapies opening across North America and Europe along with the digital and technological tools that will enable massive scale, we help people in need with a simple, evidence-based way to heal and heighten engagement with the world.
Learn more at https://www.meetfieldtrip.com, https://www.fieldtriphealth.com and https://www.fieldtriphealth.nl.
Follow us on Twitter and Instagram: @fieldtriphealth.
To receive company updates about Field Trip and to be added to the email distribution list please sign up here.
Cautionary Note Regarding Forward-Looking Information
This release includes forward-looking information (within the meaning of Canadian securities laws and within the meaning of the United States Private Securities Litigation Reform Act of 1995) regarding Field Trip and its business. Often but not always, forward-looking information can be identified by the use of words such as “expect”, “intends”, “anticipated”, “believes” or variations (including negative variations) of such words and phrases, or state that certain actions, events or results “may”, “could”, “would” or “will” be taken, occur or be achieved. Such statements are based on the current expectations and views of future events of the management of Field Trip and are based on assumptions and subject to risks and uncertainties. Although the management of Field Trip believes that the assumptions underlying these statements are reasonable, they may prove to be incorrect. The forward-looking events and circumstances discussed in this release may not occur and could differ materially as a result of known and unknown risk factors and uncertainties affecting the companies, including the timing, completion and potential outcomes of the Strategic Review, the funds available to Field Trip and the use of such funds, the ability of Field Trip to operate its clinics, the construction and commencement of construction of additional clinics, the development, patentability and viability of FT-104 and the FT-200 Group, the ability of Field Trip to complete an investigational new drug application and obtain regulatory approvals, as required, prior to initiating clinical trials for FT-104 and molecules within the FT-200 Group, the ability of Field Trip to meet eligibility requirements for clinical testing and through to more complex clinical trials, the ability of Field Trip to obtain regulatory approvals prior to each clinical trial and the ability of Field Trip to generate patient member growth, interest in the training program, interest in the KAP Co-Op Program, uptake of the KAP Co-Op Program by therapists and patients, the ability of management to sustain and continue optimization of its clinical operations, the timing and results of its research and development programs, approval of phase 1 human trials, if any, the risk that future clinical studies may not proceed as expected or may produce unfavorable results, the opening of additional clinics, the COVID-19 epidemic, the medical clinic industry, market conditions, economic factors, management’s ability to manage and to operate the business and the equity markets generally. Although Field Trip has attempted to identify important factors that could cause actual actions, events or results to differ materially from those described in forward-looking statements, there may be other factors that cause actions, events or results to differ from those anticipated, estimated or intended. Accordingly, readers should not place undue reliance on any forward-looking statements or information. No forward-looking statement can be guaranteed. Except as required by applicable securities laws, forward-looking statements speak only as of the date on which they are made and Field Trip does not undertake any obligation to publicly update or revise any forward-looking statement, whether as a result of new information, future events, or otherwise. Additional information relating to Field Trip, including its Annual Information Form, can be located on the SEDAR website at www.sedar.com and on the EDGAR section of the SEC’s website at www.sec.gov.
This press release does not constitute an offer to sell or the solicitation of an offer to buy securities.
Neither the Toronto Stock Exchange, nor its Regulation Services Provider, have approved the contents of this release or accept responsibility for the adequacy or accuracy of this release.
CONTACTS:
Media contacts:
Rachel Moskowitz
Autumn Communications
202-276-7881
press@fieldtriphealth.com
Nick Opich / McKenna Miller
KCSA Strategic Communications
212-896-1206 / 347-487-6197
press@fieldtriphealth.com
Investor contacts:
Kathleen Heaney / Tim Regan
KCSA Strategic Communications
fieldtripIR@kcsa.com
SOURCE Field Trip Health Ltd.






